Blog
EU Regulatory Insights
Regulatory updates, compliance guides, and EU market entry insights for supplement brands, cosmetic companies, and North American exporters.
France's Positive List for Botanical Ingredients: What Food Supplement Brands Must Verify Before Market Entry
France's positive list restricts botanical ingredients in food supplements. Learn what the arrêté du 24 juin 2014 requires before DGCCRF notification.
REACH Restrictions on Cosmetic Fragrances and Preservatives: Your EU Compliance Checklist for 2026
REACH restrictions directly affect fragrance and preservative ingredients in EU cosmetics. Here's the practical compliance checklist your team needs for 2026.
REACH Safety Data Sheet Requirements: What EU Suppliers and Importers Must Get Right
A practical guide to REACH SDS compliance under EU Regulation 2020/878 — 16 mandatory sections, UFI codes, SVHC disclosure, and the gaps that trigger enforcement action.
PFAS in EU Cosmetics: What Brands Must Know About the Incoming Regulation 1223/2009 Restrictions
The EU's universal PFAS restriction under REACH will reshape cosmetics compliance. Here's what Regulation 1223/2009 already covers and how to prepare for what's next.
REACH Certificate of Conformity vs. REACH Declaration: What EU Importers Actually Need
REACH CoC and REACH declarations are not the same thing — and neither substitutes for your legal obligations as an EU importer. Here's what actually matters.
REACH SVHC Compliance for Cosmetic Brands: What the Growing Candidate List Means for Your EU Formulations
With over 240 substances on the SVHC candidate list, REACH regulation compliance is reshaping EU cosmetic formulation. Here's what brands must do now.
Cosmetic Product Safety Report (CPSR): What It Is and How to Get One
A CPSR is mandatory for every cosmetic sold in the EU. This guide explains what it contains, who can write it, how long it takes, and what it costs — with a clear checklist for brands.
CPNP Notification: How to Register Your Cosmetic Product in the EU
Every cosmetic sold in the EU must be notified through the CPNP before market placement. This guide explains who submits it, what information is required, how to access the portal, and common mistakes to avoid.
EU Cosmetics Regulation 1223/2009: Complete Guide for Brands
EU Regulation 1223/2009 governs every cosmetic sold in Europe. This guide covers what it requires, who it applies to, and how to achieve compliance — including CPSR, CPNP, and responsible person obligations.

A2LA vs UKAS vs COFRAC: Which Accreditation Body Is Right for You?
Comparing A2LA, UKAS, and COFRAC — the three most important laboratory accreditation bodies for manufacturers targeting US, UK, and EU markets. Which one do you need?

Canada Natural Health Products Regulations: What US Brands Need to Know
Health Canada's Natural Health Products Regulations require a Product Licence before selling supplements in Canada. How the NHP framework works and how to get licensed.

Cosmetic Product Safety Report (CPSR): Full Requirements Guide
What is a CPSR? Who needs one? What must it contain? A complete guide to the Cosmetic Product Safety Report required under EU Cosmetics Regulation 1223/2009.

CPNP Cosmetic Notification: Step-by-Step Registration Guide
How to notify a cosmetic product through the EU CPNP portal. Step-by-step guide covering registration, required information, common errors, and timelines.
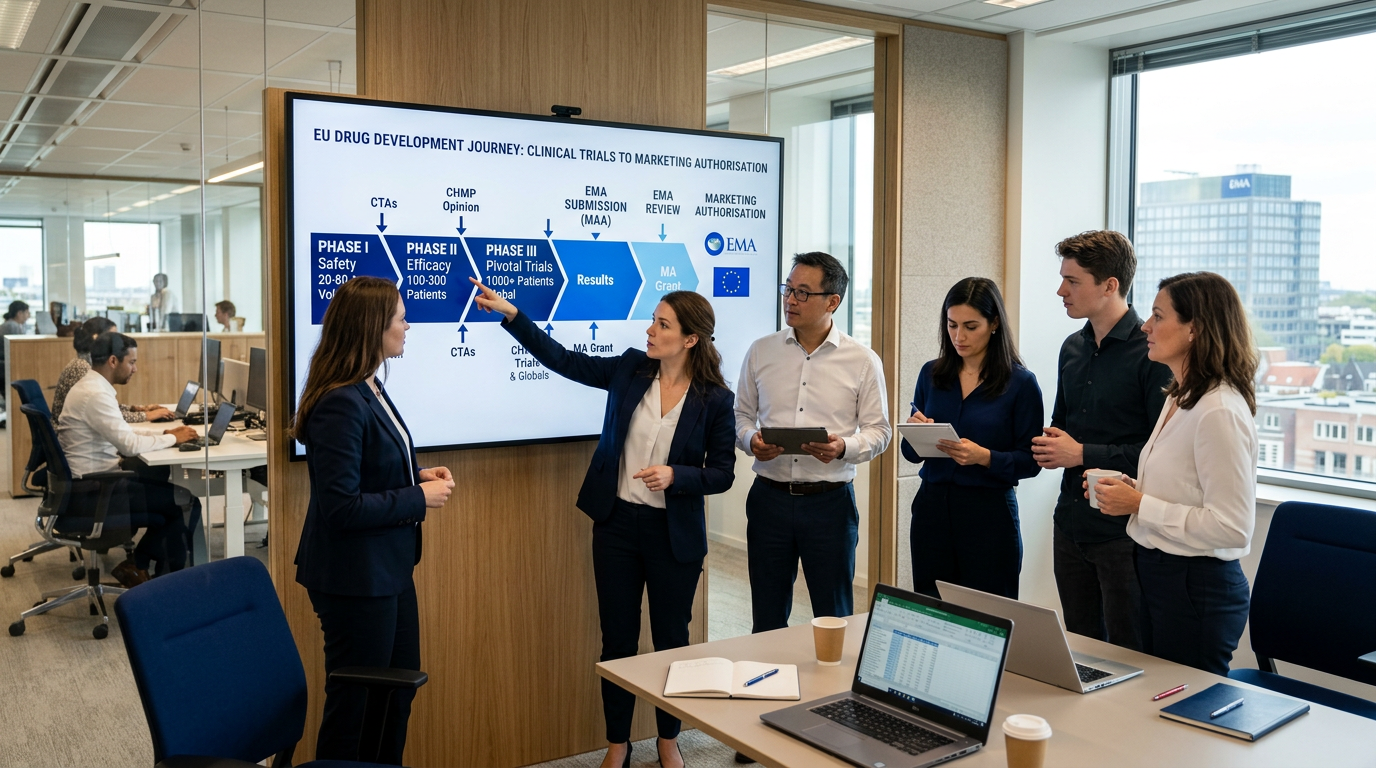
Drug Development Consulting in Europe: From IND to Marketing Authorisation
The complete EU drug development pathway from pre-clinical through marketing authorisation. Timelines, regulatory milestones, and how consulting support accelerates the process.

DSHEA vs EU Food Supplement Directive: Key Regulatory Differences
DSHEA governs supplements in the USA; EU Directive 2002/46/EC governs them in Europe. The two frameworks differ fundamentally. Here is what manufacturers need to know.

EFSA Health Claims: How to Get Your Supplement Claim Approved
EFSA evaluates health claims for food supplements under Regulation (EC) No 1924/2006. How the process works, what evidence is required, and why most claims fail.

EU Clinical Trial Regulation (EU CTR): Complete Guide for Sponsors
The EU Clinical Trial Regulation (EU) No 536/2014 replaced the 2001 Directive. This guide covers what changed, CTIS submission, and what sponsors need to know for EU trials.

EU Cosmetics Regulation 1223/2009: Prohibited and Restricted Substances
EU Cosmetics Regulation (EC) No 1223/2009 Annex II lists 1,600+ prohibited substances. Annex III lists restricted ones. What manufacturers must know about both lists.